April 5, 2022
Kavli Institute for the Physics and Mathematics of the Universe (Kavli IPMU)
 Researchers have developed a biomedical imaging system using a semiconductor detector originally developed for hard X-ray and gamma-ray space observation, and used a spectral analysis method from astronomy to take accurate images of multiple radionuclides in small animals, reports a new paper in Nature Biomedical Engineering published on April 4.
Researchers have developed a biomedical imaging system using a semiconductor detector originally developed for hard X-ray and gamma-ray space observation, and used a spectral analysis method from astronomy to take accurate images of multiple radionuclides in small animals, reports a new paper in Nature Biomedical Engineering published on April 4.
Currently, fluorescent tracers are used to image the distribution of multiple molecules in a sample under a microscope. The use of multiple tracers can reveal the distribution of numerous molecules in detail. However, when it comes to imaging molecules within the body of animals, including humans, this becomes difficult as the body tissue absorbs most of the light. So, researchers use radionuclides as probes since radiation is absorbed much less by animal tissues than optical light, allowing them to obtain images of the distribution of tracers in the body.
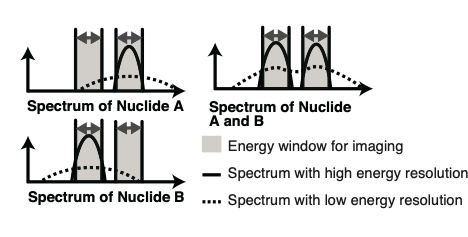
 However, current technology makes it somewhat difficult to distinguish between two or more nuclides, and has low spatial resolution. Also, it is difficult to remove noise on the image caused by radiation from other sources.
However, current technology makes it somewhat difficult to distinguish between two or more nuclides, and has low spatial resolution. Also, it is difficult to remove noise on the image caused by radiation from other sources.
To try and resolve this problem, a collaboration of researchers from fields who usually never meet one another, led by Project Assistant Professor Atsushi Yagishita from the Kavli Institute for the Physics and Mathematics of the Universe (Kavli IPMU), RIKEN, the Institute of Space and Astronautical Science, and the National Cancer Center Japan, started a project in 2018 to adapt technology used in space observation for medical research.
Their latest study highlights two achievements.
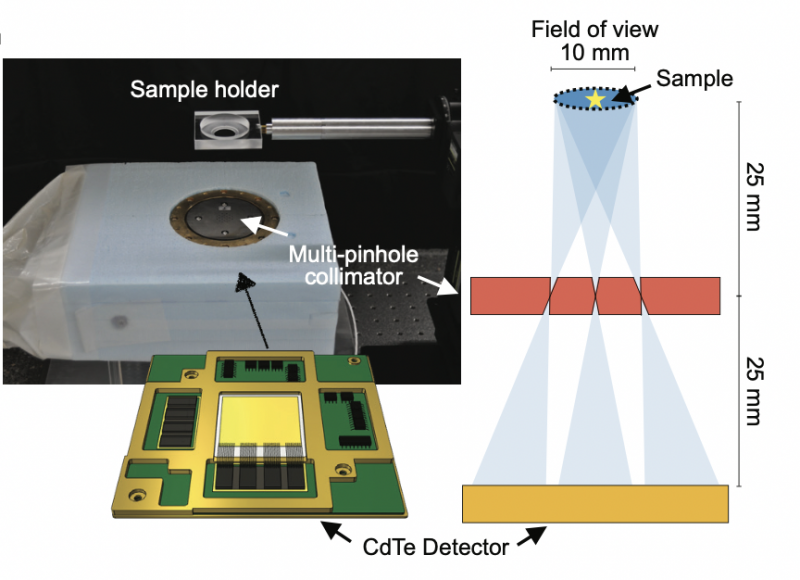
One of the team’s achievements was the development of an imager, named IPMU imager, which was equipped with a cadmium telluride semiconductor detector with high energy resolution, meaning it had the ability to distinguish radiations of different energies, and a multi-pinhole collimator that could achieve high spatial resolution for radionuclide imaging. The IPMU imager made it easy to distinguish between radiations with different energies.
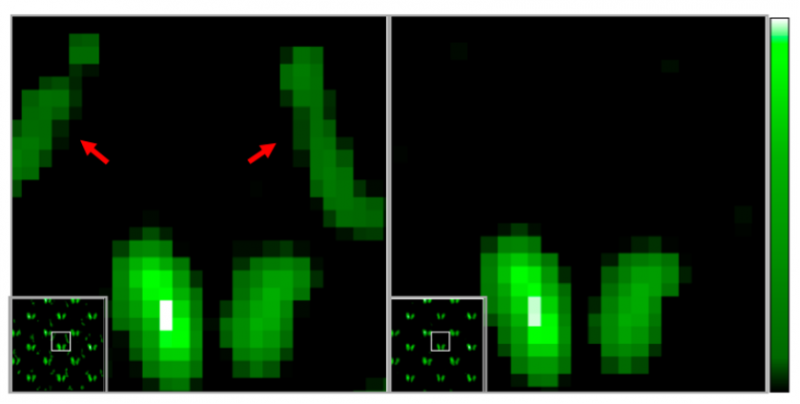
 Still, there was room for improvement. The IPMU Imager could not eliminate all noise, especially when two radionuclides released similar radioactive emissions.
Still, there was room for improvement. The IPMU Imager could not eliminate all noise, especially when two radionuclides released similar radioactive emissions.
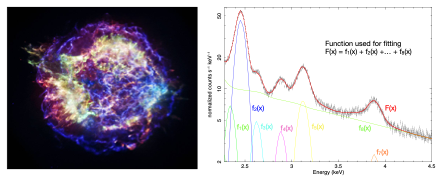
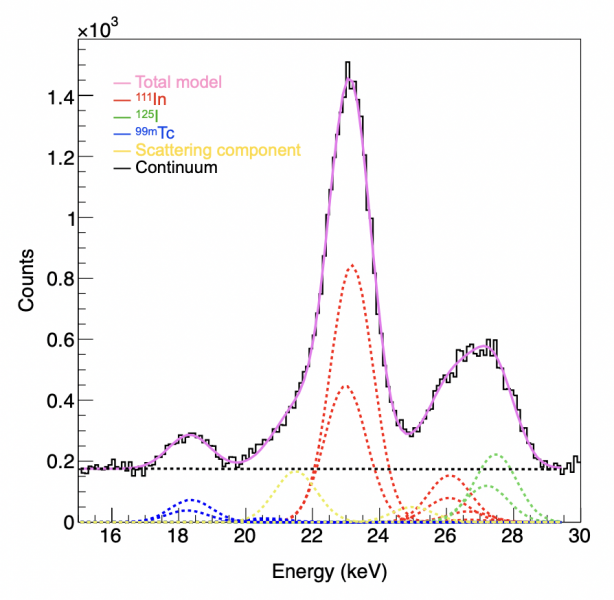
So the team’s second achievement was to use “fitting”, a spectroscopic X-ray analysis method used in the field of astronomy. What they were able to find was that using the technique allowed them to identify the source of all radiation. This eliminated noise radiation and made it possible to obtain accurate images using only radiation from the target nuclide.
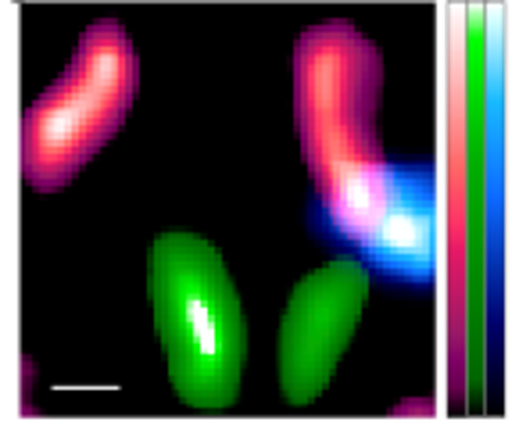
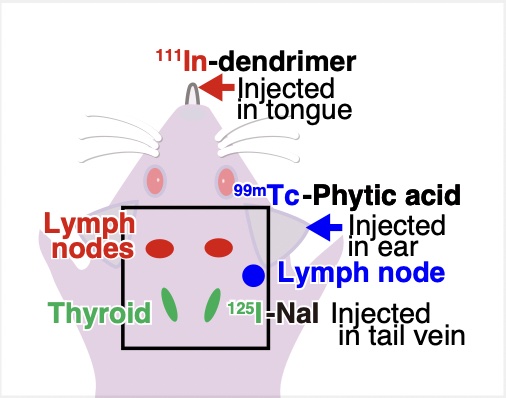
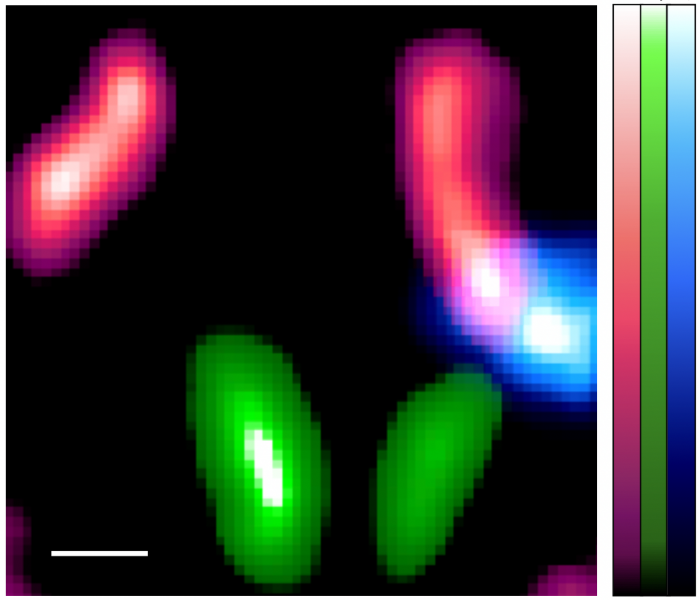
Once the researchers were confident with their technique, they moved onto confirming it using mice. Three types of tracers with technetium-99m (99mTc), Indium-111 (111In), and Iodine-125 (125I), respectively, were used in the experiment. These tracers accumulated in the lymph nodes and thyroid, respectively. Raw images showed noise and ghosting caused by radiation from other sources. However, by using the fitting technique, the researchers were able to identify unwanted radiation sources. When imaging 125I, only the thyroid gland where 125I accumulates could be accurately and finely delineated by eliminating background noise and ghosting.
The researchers say there is still a way to go. While this study was an experiment involving a SPECT prototype, the researchers have already developed a full spec SPECT imaging device.
The team’s method can be applied to biomedical research, radiopharmaceutical development, and clinical diagnostic techniques.
Paper details
Journal: Nature Biomedical Engineering
Title: Simultaneous visualization of multiple radionuclides in vivo
Authors: Atsushi Yagishita (1,2), Shin’ichiro Takeda (1), Miho Katsuragawa (1), Tenyo Kawamura (3,1), Hideaki Matsumura (1), Tadashi Orita (1), Izumi O. Umeda (1), Goro Yabu (3,1), Pietro Caradonna (1), Tadayuki Takahashi (1), Shin Watanabe (4,1), Yousuke Kanayama (5), Hiroshi Mizuma (5), Kazunobu Ohnuki (2), Hirofumi Fujii (2)
Author affiliations:
1. Kavli Institute for the Physics and Mathematics of the Universe (Kavli IPMU, WPI), The University of Tokyo, 5-1-5, Kashiwanoha, Kashiwa, Chiba 277-8583, Japan
2. Exploratory Oncology Research and Clinical Trial Center, National Cancer Center, 6-5-1 Kashiwanoha, Kashiwa,Chiba 277-8577, Japan
3. Department of Physics, The University of Tokyo, 7-3-1 Hongo, Bunkyo, Tokyo 113-0033, Japan
4. Institute of Space and Astronautical Science (ISAS)/JAXA, 3-1-1 Yoshinodai, Chuo-ku, Sagamihara, Kanagawa 252-5210, Japan
5. Laboratory for Pathophysiological and Health Science, RIKEN Center for Biosystems Dynamics Research, 6-7-3 Minatojima-minamimachi, Chuo-ku, Kobe, Hyogo 650-0047, Japan
DOI: 10.1038/s41551-022-00866-6 (published 4 April, 2022)
Paper abstract URL (Nature Biomedical Engineering)
Media contact
Motoko Kakubayashi
Press officer
Kavli Institute for the Physics and Mathematics of the Universe (Kavli IPMU), The University of Tokyo
E-mail:press_at_ipmu.jp
* Please change _at_ to @